Give back to your high performer by treating arthritis.

Osteoarthritis is the degeneration of cartilage in the joint. It is often due to normal “wear and tear” over time but can develop following a single traumatic event.1
Horses with osteoarthritis may exhibit:1
Owners may recognize common early signs, such as:
Hyaluronic acid is a naturally occurring component of synovial fluid in joints, which acts as a lubricant and shock absorber. Synovial fluid promotes smooth joint movement and reduces friction between cartilage surfaces.
Hyaluronate sodium (a form of hyaluronic acid) exerts an anti-inflammatory action, which can help calm synovial inflammation associated with equine osteoarthritis.2 Hyaluronic acid also provides lubrication of joints.3
Intravenous joint injections of hyaluronic acid are FDA approved to help alleviate the signs of lameness and have been a trusted treatment for equine osteoarthritis for nearly 30 years.4
Hyaluronic acid is a naturally occurring substance present in the connective tissue in all mammals.2
It is found in many tissues and fluids, but more abundantly in articular cartilage and synovial fluid.4

Hyaluronate sodium exerts an anti-inflammatory action, which can help calm synovial inflammation associated with equine osteoarthritis.2 Hyaluronic acid also provides lubrication of joints.4

NexHA is FDA approved as bioequivalent to Legend IV at an affordable price.
Trust generic products to be produced under the same standards and have demonstrated bioequivalence to the brand-name product.5
Using affordable generics allows owners to invest in other important veterinary services.
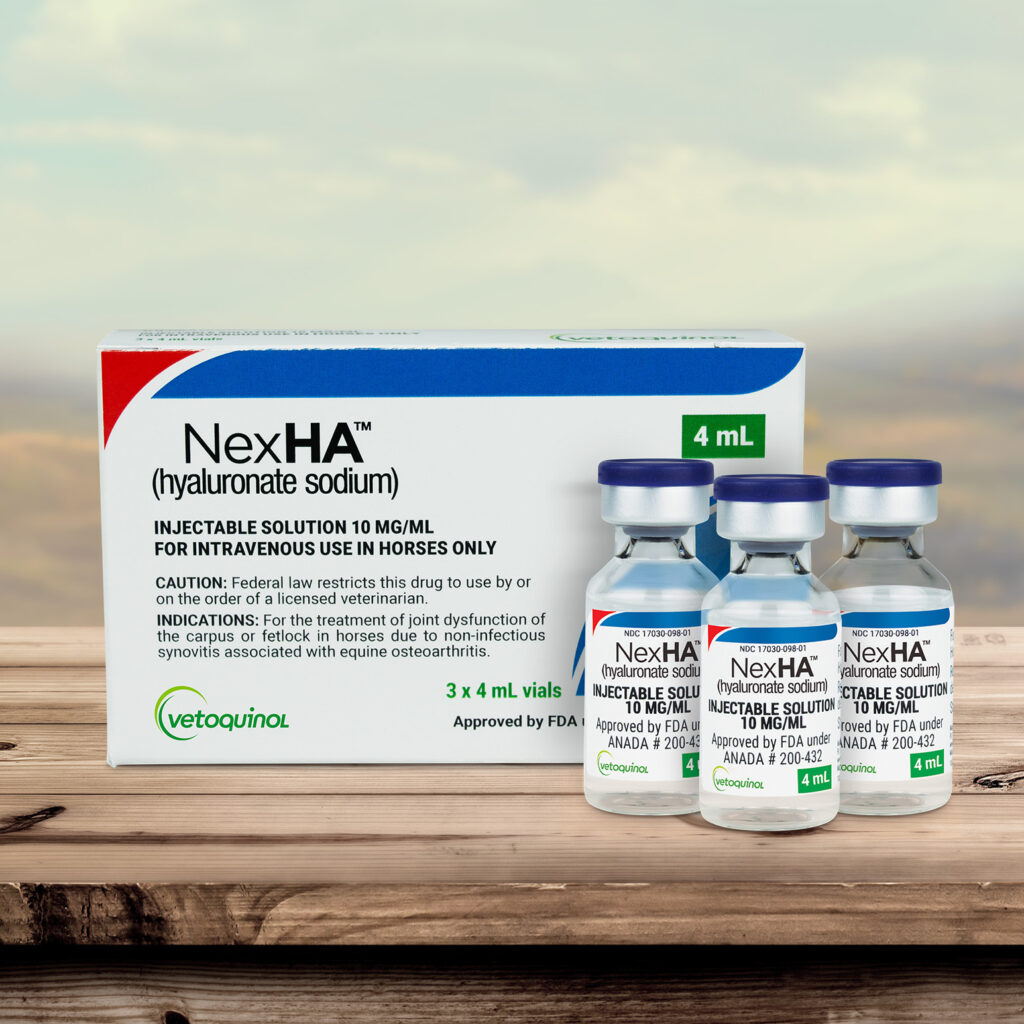
NexHA is approved for the treatment of joint dysfunction of the carpus or fetlock in horses due to non-infectious synovitis associated with equine osteoarthritis.

In a study, overall clinical improvement was judged as excellent or good in 90% of the cases treated intravenously.2


In one study, lameness improved for about six weeks after intravenous administration of sodium hyaluronate.6

Treatment with NexHA may be repeated at weekly intervals for a total of three treatments. The product is available in a 4 mL (40 mg) vial size for intravenous injection. NexHA can be used with other joint therapies.
NexHA is made by extracting hyaluronic acid from the capsule of Streptococcus spp., which is then purified, resulting in a form that is essentially free of protein and nucleic acids. The hyaluronic acid produced by bacteria is the same structure and configuration as found in mammals.2
NexHA is pyrogen free, sterile, and does not contain a preservative.
Save up to $85 for a 3-shot protocol with NexHA compared to the name brand!
Veterinarians can purchase NexHA from their Vetoquinol Territory Manager or preferred distributor representative. NexHA is a prescription-only product.
Owners with a prescription can order NexHA from their preferred online veterinary pharmacy.
NexHA is available in a 3-vial box, which is the perfect size for a complete treatment!
Frequently Asked Questions
What is NexHA for horses?
NexHA is an FDA-approved intravenous (IV) treatment for equine osteoarthritis. It is FDA approved as bioequivalent to Legend IV.
Can NexHA be used with other joint therapies?
Yes! NexHA can be used with other joint therapies.
How long does it take NexHA to work?
In one study, lameness improved for about six weeks after intravenous administration of sodium hyaluronate.6
How much hyaluronic acid do horses need?
Use 4 mL (40 mg) NexHA injected intravenously. Treatment with NexHA may be repeated at weekly intervals for a total of three treatments.
What are the possible side effects?
While all adverse reactions are not reported, the following adverse reactions are based on voluntary post-approval reporting for hyaluronate sodium injectable solution: Occasional depression, lethargy, and fever. For full prescribing information, visit www.vetoquinolusa.com/nexha-info
Important Safety Information
NexHA™ (hyaluronate sodium) Injectable Solution is indicated in the treatment of joint dysfunction of the carpus or fetlock in horses due to non-infectious synovitis associated with equine osteoarthritis.
IMPORTANT SAFETY INFORMATION:
Federal law restricts this drug to use by or on the order of a licensed veterinarian. For use only in horses. Do not use in horses intended for human consumption.
NexHA™ (hyaluronate sodium) Injectable Solution 4 mL is approved only for intravenous use. The safety of NexHA™ (hyaluronate sodium) Injectable Solution has not been evaluated in breeding stallions or in breeding, pregnant or lactating mares. Not for use in humans. Keep this and all other drugs out of reach of children. Post- Approval Experience: While all adverse reactions are not reported, the following adverse reactions are based on voluntary post-approval reporting for hyaluronate sodium injectable solution: Occasional depression, lethargy and fever. For full prescribing information, visit www.vetoquinolusa.com/nexha-info.
REFERENCES: